Journal of Traditional Chinese Medicine ›› 2022, Vol. 42 ›› Issue (4): 622-632.DOI: 10.19852/j.cnki.jtcm.20220617.002
• Research Articles • Previous Articles Next Articles
Identification of novel biomarkers and therapeutic target candidates for stasis-heat symptom pattern of acute intracerebral hemorrhage by quantitative plasma proteomics
WEI Lexin1, LI Weiyi1, TIAN Ting2, ZHANG Ning1, YANG Shijing1, YANG Dongqing1, LI Guochun1( ), YE Fang3(
), YE Fang3( )
)
- 1 Department of Public Health, School of Medicine and Holistic Integrative Medicine, Nanjing University of Chinese Medicine, Nanjing 210023, China
2 Emergency Department, Nanjing Hospital of Chinese Medicine affiliated to Nanjing University of Chinese Medicine, Nanjing 210001, China
3 the Key Laboratory Department of Stasis-heat Pathogenesis of Traditional Chinese Medicine, the First Clinical Medical College, Nanjing University of Chinese Medicine, Nanjing 210001, China
-
Received:2021-04-22Accepted:2021-07-18Online:2022-08-15Published:2022-07-12 -
Contact:LI Guochun,YE Fang -
About author:Prof. YE Fang, the Key Research Department of Stasis-heat Pathogenesis of Traditional Chinese Medicine, the First Clinical Medical College, Nanjing University of Chinese Medicine, Nanjing 210023, China. 260958@njucm.edu.cn, Telephone: +86-25-85511926
Prof. LI Guochun, Department of Public Health, School of Medicine and Holistic Integrative Medicine, Nanjing University of Chinese Medicine, Nanjing 210023, China. dr.guochun_li@njucm.edu.cn;
-
Supported by:Construction and Application of Decision Support System of TCM Master Zhou Zhongying's Experience Learning in Distinguishing and Treating Major Diseases based on the Intelligent Technology(BE2019723);Dynamic Measurement and Study of Biomarker on Pathogenic Unit of Stasis-Heat in Acute Intracerebral Hemorrhage(813735)
Cite this article
WEI Lexin, LI Weiyi, TIAN Ting, ZHANG Ning, YANG Shijing, YANG Dongqing, LI Guochun, YE Fang. Identification of novel biomarkers and therapeutic target candidates for stasis-heat symptom pattern of acute intracerebral hemorrhage by quantitative plasma proteomics[J]. Journal of Traditional Chinese Medicine, 2022, 42(4): 622-632.
share this article
| Block | Tissue | Disease or normal | Patient number | Age/ gender | Score of stasis-heat | District and hospital | TMT labeling reagent ID | TMT labeling code |
|---|---|---|---|---|---|---|---|---|
| 1 | 1 | Normal | 1 | 66/F | 0 | Nanjing | 1 | 126 |
| 1 | 2 | AICH (stasis-heat) | 2 | 61/F | 14 | Nanjing | 1 | 127 |
| 1 | 4 | AICH (non-stasis-heat) | 3 | 65/F | 7 | Nanjing | 1 | 129 |
| Reference | - | - | - | - | 1 | 131 | ||
| 2 | 6 | Normal | 4 | 60/M | 0 | Nanjing | 2 | 126 |
| 2 | 7 | AICH (stasis-heat) | 5 | 56/M | 21 | Nanjing | 2 | 127 |
| 2 | 9 | AICH (non-stasis-heat) | 6 | 58/M | 9 | Nanjing | 2 | 129 |
| Reference | - | - | - | - | 2 | 131 | ||
| 3 | 11 | Normal | 7 | 71/M | 0 | Zhenjiang | 3 | 126 |
| 3 | 12 | AICH (stasis-heat) | 8 | 69/M | 14 | Zhenjiang | 3 | 127 |
| 3 | 14 | AICH (non-stasis-heat) | 9 | 66/M | 9 | Zhenjiang | 3 | 129 |
| Reference | - | - | - | - | 3 | 131 | ||
| 4 | 16 | Normal | 10 | 56/F | 0 | Zhenjiang | 4 | 126 |
| 4 | 17 | AICH (stasis-heat) | 11 | 61/F | 13 | Zhenjiang | 4 | 127 |
| 4 | 19 | AICH (non-stasis-heat) | 12 | 57/F | 4 | Zhenjiang | 4 | 129 |
| Reference | - | - | - | - | 4 | 131 |
Table 1 Experimental protocol and basic characteristics in AICH patients with stasis-heat, AICH patients with non-stasis-heat, and health controls groups (n = 4 per group)
| Block | Tissue | Disease or normal | Patient number | Age/ gender | Score of stasis-heat | District and hospital | TMT labeling reagent ID | TMT labeling code |
|---|---|---|---|---|---|---|---|---|
| 1 | 1 | Normal | 1 | 66/F | 0 | Nanjing | 1 | 126 |
| 1 | 2 | AICH (stasis-heat) | 2 | 61/F | 14 | Nanjing | 1 | 127 |
| 1 | 4 | AICH (non-stasis-heat) | 3 | 65/F | 7 | Nanjing | 1 | 129 |
| Reference | - | - | - | - | 1 | 131 | ||
| 2 | 6 | Normal | 4 | 60/M | 0 | Nanjing | 2 | 126 |
| 2 | 7 | AICH (stasis-heat) | 5 | 56/M | 21 | Nanjing | 2 | 127 |
| 2 | 9 | AICH (non-stasis-heat) | 6 | 58/M | 9 | Nanjing | 2 | 129 |
| Reference | - | - | - | - | 2 | 131 | ||
| 3 | 11 | Normal | 7 | 71/M | 0 | Zhenjiang | 3 | 126 |
| 3 | 12 | AICH (stasis-heat) | 8 | 69/M | 14 | Zhenjiang | 3 | 127 |
| 3 | 14 | AICH (non-stasis-heat) | 9 | 66/M | 9 | Zhenjiang | 3 | 129 |
| Reference | - | - | - | - | 3 | 131 | ||
| 4 | 16 | Normal | 10 | 56/F | 0 | Zhenjiang | 4 | 126 |
| 4 | 17 | AICH (stasis-heat) | 11 | 61/F | 13 | Zhenjiang | 4 | 127 |
| 4 | 19 | AICH (non-stasis-heat) | 12 | 57/F | 4 | Zhenjiang | 4 | 129 |
| Reference | - | - | - | - | 4 | 131 |

Figure 1 Experimental profile AICH: acute intracerebral hemorrhage; SH: stasis-heat; NSH: non-stasis-heat; HC: healthy controls; TMT: tandem mass tags; LC-MS/MS: liquid chromatography tandem-mass spectrometry.
| No. | UniProt accession | Symbol | Entrez gene name | Abundance in SHP | Abundance in HG | Fold change | 95%CI for fold change | P value | |
|---|---|---|---|---|---|---|---|---|---|
| 1 | V9HWD8 | A1BG | Alpha-1-B glycoprotein | 1.678±0.102 | 1.127±0.359 | 1.490±0.091 | 1.490 (1.346, 1.634) | 0.034 | |
| 2 | A0A024R694 | ACTN1 | Actinin, alpha 1 | 1.408±0.086 | 0.680±0.238 | 2.069±0.126 | 2.069 (0.939, 3.200) | 0.051 | |
| 3 | P06727 | APOA4 | Apolipoprotein A-IV | 0.772±0.078 | 1.418±0.334 | -1.851±0.186 | -1.851 (-2.147, -1.554) | 0.004 | |
| 4 | V9HWE3 | CA1 | Carbonic anhydrase I | 1.015±0.413 | 0.667±0.292 | 1.002±1.586 | 1.002 (-1.521, 3.525) | 0.016 | |
| 5 | V9HW21 | CA2 | Carbonic anhydrase II | 1.366±0.138 | 0.901±0.071 | 1.516±0.153 | 1.516 (1.136, 1.896) | 0.022 | |
| 6 | A0A087WXI2 | FCGBP | Fc fragment of igg binding protein | 1.076±0.095 | 0.801±0.180 | 1.343±0.118 | 1.343 (1.155, 1.531) | 0.047 | |
| 7 | P02671 | FGA | Fibrinogen alpha chain | 1.151±0.144 | 0.694±0.114 | 1.658±0.208 | 1.658 (1.327, 1.989) | 0.043 | |
| 8 | V9HVY1 | FGB | Fibrinogen beta chain | 1.069±0.042 | 0.719±0.145 | 1.487±0.058 | 1.487 (1.394, 1.580) | 0.042 | |
| 9 | V9HWI6 | GC | Group-specific component (vitamin D binding protein) | 0.943±0.155 | 1.553±0.497 | -1.688±0.327 | -1.688 (-2.209, -1.167) | 0.065 | |
| 10 | A8K335 | GGH | Gamma-glutamyl hydrolase (conjugase, folylpolygammaglutamyl hydrolase) | 1.093±0.232 | 0.723±0.138 | 1.513±0.321 | 1.513 (0.716, 2.310) | 0.054 | |
| 11 | P06396 | GSN | Gelsolin | 0.974±0.092 | 1.367±0.326 | -1.414±0.134 | -1.414 (-1.628, -1.200) | 0.049 | |
| 12 | D9YZU5 | HBB | Hemoglobin, beta | 1.201±0.741 | 0.511±0.205 | 1.782±2.468 | 1.782 (-2.145, 5.709) | 0.056 | |
| 13 | Q7Z379 | IGH | Immunoglobulin heavy locus | 0.866±0.424 | 1.889±0.614 | -2.504±0.998 | -2.504 (-4.982, -0.026) | 0.067 | |
| 14 | P18428 | LBP | Lipopolysaccharide binding protein | 1.108±0.258 | 0.610±0.167 | 1.583±0.369 | 1.583 (0.996, 2.170) | 0.067 | |
| 15 | V9HWF6 | ORM1 | Orosomucoid 1 | 0.837±0.097 | 0.472±0.121 | 1.774±0.206 | 1.774 (1.446, 2.102) | 0.031 | |
| 16 | P07737 | PFN1 | Profilin 1 | 1.276±0.326 | 0.758±0.238 | 1.682±0.410 | 1.682 (0.998, 2.366) | 0.022 | |
| 17 | D3JV41 | PPBP | Pro-platelet basic protein (chemokine (C-X-C motif) ligand 7) | 1.433±0.508 | 0.627±0.095 | 2.284±0.810 | 2.284 (0.995, 3.574) | 0.041 | |
| 18 | B4DF70 | PRDX2 | Peroxiredoxin 2 | 1.423±0.209 | 0.735±0.111 | 1.937±0.285 | 1.937 (1.230, 2.645) | 0.009 | |
| 19 | A8K2W3 | SDPR | Serum deprivation response | 1.066±0.332 | 0.449±0.068 | 2.377±0.741 | 2.377 (0.538, 4.217) | 0.053 | |
| 20 | A2VCK8 | TMSB10/TMSB4X | Thymosin beta 10 | 1.762±0.221 | 0.634±0.262 | 2.778±0.349 | 2.778 (2.223, 3.333) | 0.029 | |
| 21 | A0A024QZN4 | VCL | Vinculin | 1.369±0.125 | 0.852±0.141 | 1.607±0.146 | 1.607 (1.375, 1.840) | 0.015 | |
Table 2 List of proteins with different abundance in stasis-heat patients compared with health group ($\bar{x}±s$)
| No. | UniProt accession | Symbol | Entrez gene name | Abundance in SHP | Abundance in HG | Fold change | 95%CI for fold change | P value | |
|---|---|---|---|---|---|---|---|---|---|
| 1 | V9HWD8 | A1BG | Alpha-1-B glycoprotein | 1.678±0.102 | 1.127±0.359 | 1.490±0.091 | 1.490 (1.346, 1.634) | 0.034 | |
| 2 | A0A024R694 | ACTN1 | Actinin, alpha 1 | 1.408±0.086 | 0.680±0.238 | 2.069±0.126 | 2.069 (0.939, 3.200) | 0.051 | |
| 3 | P06727 | APOA4 | Apolipoprotein A-IV | 0.772±0.078 | 1.418±0.334 | -1.851±0.186 | -1.851 (-2.147, -1.554) | 0.004 | |
| 4 | V9HWE3 | CA1 | Carbonic anhydrase I | 1.015±0.413 | 0.667±0.292 | 1.002±1.586 | 1.002 (-1.521, 3.525) | 0.016 | |
| 5 | V9HW21 | CA2 | Carbonic anhydrase II | 1.366±0.138 | 0.901±0.071 | 1.516±0.153 | 1.516 (1.136, 1.896) | 0.022 | |
| 6 | A0A087WXI2 | FCGBP | Fc fragment of igg binding protein | 1.076±0.095 | 0.801±0.180 | 1.343±0.118 | 1.343 (1.155, 1.531) | 0.047 | |
| 7 | P02671 | FGA | Fibrinogen alpha chain | 1.151±0.144 | 0.694±0.114 | 1.658±0.208 | 1.658 (1.327, 1.989) | 0.043 | |
| 8 | V9HVY1 | FGB | Fibrinogen beta chain | 1.069±0.042 | 0.719±0.145 | 1.487±0.058 | 1.487 (1.394, 1.580) | 0.042 | |
| 9 | V9HWI6 | GC | Group-specific component (vitamin D binding protein) | 0.943±0.155 | 1.553±0.497 | -1.688±0.327 | -1.688 (-2.209, -1.167) | 0.065 | |
| 10 | A8K335 | GGH | Gamma-glutamyl hydrolase (conjugase, folylpolygammaglutamyl hydrolase) | 1.093±0.232 | 0.723±0.138 | 1.513±0.321 | 1.513 (0.716, 2.310) | 0.054 | |
| 11 | P06396 | GSN | Gelsolin | 0.974±0.092 | 1.367±0.326 | -1.414±0.134 | -1.414 (-1.628, -1.200) | 0.049 | |
| 12 | D9YZU5 | HBB | Hemoglobin, beta | 1.201±0.741 | 0.511±0.205 | 1.782±2.468 | 1.782 (-2.145, 5.709) | 0.056 | |
| 13 | Q7Z379 | IGH | Immunoglobulin heavy locus | 0.866±0.424 | 1.889±0.614 | -2.504±0.998 | -2.504 (-4.982, -0.026) | 0.067 | |
| 14 | P18428 | LBP | Lipopolysaccharide binding protein | 1.108±0.258 | 0.610±0.167 | 1.583±0.369 | 1.583 (0.996, 2.170) | 0.067 | |
| 15 | V9HWF6 | ORM1 | Orosomucoid 1 | 0.837±0.097 | 0.472±0.121 | 1.774±0.206 | 1.774 (1.446, 2.102) | 0.031 | |
| 16 | P07737 | PFN1 | Profilin 1 | 1.276±0.326 | 0.758±0.238 | 1.682±0.410 | 1.682 (0.998, 2.366) | 0.022 | |
| 17 | D3JV41 | PPBP | Pro-platelet basic protein (chemokine (C-X-C motif) ligand 7) | 1.433±0.508 | 0.627±0.095 | 2.284±0.810 | 2.284 (0.995, 3.574) | 0.041 | |
| 18 | B4DF70 | PRDX2 | Peroxiredoxin 2 | 1.423±0.209 | 0.735±0.111 | 1.937±0.285 | 1.937 (1.230, 2.645) | 0.009 | |
| 19 | A8K2W3 | SDPR | Serum deprivation response | 1.066±0.332 | 0.449±0.068 | 2.377±0.741 | 2.377 (0.538, 4.217) | 0.053 | |
| 20 | A2VCK8 | TMSB10/TMSB4X | Thymosin beta 10 | 1.762±0.221 | 0.634±0.262 | 2.778±0.349 | 2.778 (2.223, 3.333) | 0.029 | |
| 21 | A0A024QZN4 | VCL | Vinculin | 1.369±0.125 | 0.852±0.141 | 1.607±0.146 | 1.607 (1.375, 1.840) | 0.015 | |
| No. | UniProt accession | Symbol | Entrez gene name | Abundance in nonSHP | Abundance in HG | Fold change | 95%CI for fold change | P value |
|---|---|---|---|---|---|---|---|---|
| 1 | P06727 | APOA4 | Apolipoprotein A-IV | 0.820±0.152 | 1.418±0.334 | –1.781±0.386 | –1.77(–2.39,–1.16) | 0.006 |
| 2 | V9HW21 | CA2 | Carbonic anhydrase II | 1.225±0.267 | 0.901±0.070 | 1.360±0.296 | 1.35(0.62,2.09) | 0.073 |
| 3 | H0YCV9 | CD44 | CD44 molecule (Indian blood group) | 1.183±0.641 | 0.944±0.671 | 1.252±0.679 | 1.25(0.17,2.33) | 0.003 |
| 4 | L8EB32 | COL14A1 | Collagen, type XIV, alpha 1 | 0.994±0.138 | 2.573±0.453 | –2.614±0.365 | –2.61(–5.89,0.66) | 0.087 |
| 5 | Q53S54 | CUL3 | Cullin 3 | 1.406±0.472 | 0.496±0.144 | 2.831±0.950 | 2.82(0.46,5.19) | 0.063 |
| 6 | V9HWI6 | GC | Group-specific component (vitamin D binding protein) | 0.980±0.300 | 1.552±0.496 | –1.679±0.422 | –1.67(–2.35,–1.00) | 0.083 |
| 7 | P06396 | GSN | Gelsolin | 0.919±0.105 | 1.367±0.325 | –1.501±0.176 | –1.50(–1.78,–1.22) | 0.029 |
| 8 | Q6ZW64 | IGH | Immunoglobulin heavy locus | 1.216±0.404 | 0.528±0.238 | 2.304±0.765 | 2.30(1.08,3.52) | 0.064 |
| 9 | A0A087X0P6 | IGKV2D-29 | Immunoglobulin kappa variable 2D-29 | 0.659±0.188 | 1.133±0.032 | –1.791±0.512 | –1.79(–6.39,2.81) | 0.081 |
| 10 | P18428 | LBP | Lipopolysaccharide binding protein | 1.252±0.377 | 0.699±0.167 | 1.789±0.539 | 1.78(0.93,2.64) | 0.018 |
| 11 | V9HWF6 | ORM1 | Orosomucoid 1 | 0.985±0.323 | 0.471±0.120 | 2.090±0.686 | 2.08(0.99,3.18) | 0.006 |
Table 3 List of proteins with different abundance in non-stasis heat patients compared with health group ($\bar{x}±s$)
| No. | UniProt accession | Symbol | Entrez gene name | Abundance in nonSHP | Abundance in HG | Fold change | 95%CI for fold change | P value |
|---|---|---|---|---|---|---|---|---|
| 1 | P06727 | APOA4 | Apolipoprotein A-IV | 0.820±0.152 | 1.418±0.334 | –1.781±0.386 | –1.77(–2.39,–1.16) | 0.006 |
| 2 | V9HW21 | CA2 | Carbonic anhydrase II | 1.225±0.267 | 0.901±0.070 | 1.360±0.296 | 1.35(0.62,2.09) | 0.073 |
| 3 | H0YCV9 | CD44 | CD44 molecule (Indian blood group) | 1.183±0.641 | 0.944±0.671 | 1.252±0.679 | 1.25(0.17,2.33) | 0.003 |
| 4 | L8EB32 | COL14A1 | Collagen, type XIV, alpha 1 | 0.994±0.138 | 2.573±0.453 | –2.614±0.365 | –2.61(–5.89,0.66) | 0.087 |
| 5 | Q53S54 | CUL3 | Cullin 3 | 1.406±0.472 | 0.496±0.144 | 2.831±0.950 | 2.82(0.46,5.19) | 0.063 |
| 6 | V9HWI6 | GC | Group-specific component (vitamin D binding protein) | 0.980±0.300 | 1.552±0.496 | –1.679±0.422 | –1.67(–2.35,–1.00) | 0.083 |
| 7 | P06396 | GSN | Gelsolin | 0.919±0.105 | 1.367±0.325 | –1.501±0.176 | –1.50(–1.78,–1.22) | 0.029 |
| 8 | Q6ZW64 | IGH | Immunoglobulin heavy locus | 1.216±0.404 | 0.528±0.238 | 2.304±0.765 | 2.30(1.08,3.52) | 0.064 |
| 9 | A0A087X0P6 | IGKV2D-29 | Immunoglobulin kappa variable 2D-29 | 0.659±0.188 | 1.133±0.032 | –1.791±0.512 | –1.79(–6.39,2.81) | 0.081 |
| 10 | P18428 | LBP | Lipopolysaccharide binding protein | 1.252±0.377 | 0.699±0.167 | 1.789±0.539 | 1.78(0.93,2.64) | 0.018 |
| 11 | V9HWF6 | ORM1 | Orosomucoid 1 | 0.985±0.323 | 0.471±0.120 | 2.090±0.686 | 2.08(0.99,3.18) | 0.006 |
| No. | UniProt accession | Symbol | Entrez gene name | Abundance in SHP | Abundance in nonSHP | Fold change | 95%CI for fold change | P value | |
|---|---|---|---|---|---|---|---|---|---|
| 1 | V9HWD8 | A1BG | Alpha-1-B glycoprotein | 1.678±0.102 | 1.017±0.130 | 1.648±0.100 | 1.64(1.48,1.80) | 0.015 | |
| 2 | A0A024R694 | ACTN1 | Actinin, alpha 1 | 1.407±0.085 | 0.750±0.060 | 1.876±0.114 | 1.87(0.85,2.90) | 0.062 | |
| 3 | V9HWE3 | CA1 | Carbonic anhydrase I | 1.015±0.412 | 0.761±0.232 | 1.333±0.542 | 1.33(0.47,2.19) | 0.061 | |
| 4 | H0YCV9 | CD44 | CD44 molecule (Indian blood group) | 1.062±0.615 | 1.183±0.641 | –1.149±1.375 | –1.15(–3.33,1.03) | 0.074 | |
| 5 | A5PL27 | CP | Ceruloplasmin (ferroxidase) | 1.308±0.115 | 0.941±0.080 | 1.390±0.123 | 1.38(1.19,1.58) | 0.046 | |
| 6 | B4DF70 | PRDX2 | Peroxiredoxin 2 | 1.422±0.209 | 0.921±0.136 | 1.543±0.226 | 1.54(0.98,2.10) | 0.029 | |
| 7 | A0A024QZN4 | VCL | Vinculin | 1.369±0.124 | 1.025±0.242 | 1.334±0.121 | 1.33(1.14,1.52) | 0.087 | |
Table 4 List of proteins with different abundance in stasis-heat patients compared with non-stasis-heat patients ($\bar{x}±s$)
| No. | UniProt accession | Symbol | Entrez gene name | Abundance in SHP | Abundance in nonSHP | Fold change | 95%CI for fold change | P value | |
|---|---|---|---|---|---|---|---|---|---|
| 1 | V9HWD8 | A1BG | Alpha-1-B glycoprotein | 1.678±0.102 | 1.017±0.130 | 1.648±0.100 | 1.64(1.48,1.80) | 0.015 | |
| 2 | A0A024R694 | ACTN1 | Actinin, alpha 1 | 1.407±0.085 | 0.750±0.060 | 1.876±0.114 | 1.87(0.85,2.90) | 0.062 | |
| 3 | V9HWE3 | CA1 | Carbonic anhydrase I | 1.015±0.412 | 0.761±0.232 | 1.333±0.542 | 1.33(0.47,2.19) | 0.061 | |
| 4 | H0YCV9 | CD44 | CD44 molecule (Indian blood group) | 1.062±0.615 | 1.183±0.641 | –1.149±1.375 | –1.15(–3.33,1.03) | 0.074 | |
| 5 | A5PL27 | CP | Ceruloplasmin (ferroxidase) | 1.308±0.115 | 0.941±0.080 | 1.390±0.123 | 1.38(1.19,1.58) | 0.046 | |
| 6 | B4DF70 | PRDX2 | Peroxiredoxin 2 | 1.422±0.209 | 0.921±0.136 | 1.543±0.226 | 1.54(0.98,2.10) | 0.029 | |
| 7 | A0A024QZN4 | VCL | Vinculin | 1.369±0.124 | 1.025±0.242 | 1.334±0.121 | 1.33(1.14,1.52) | 0.087 | |
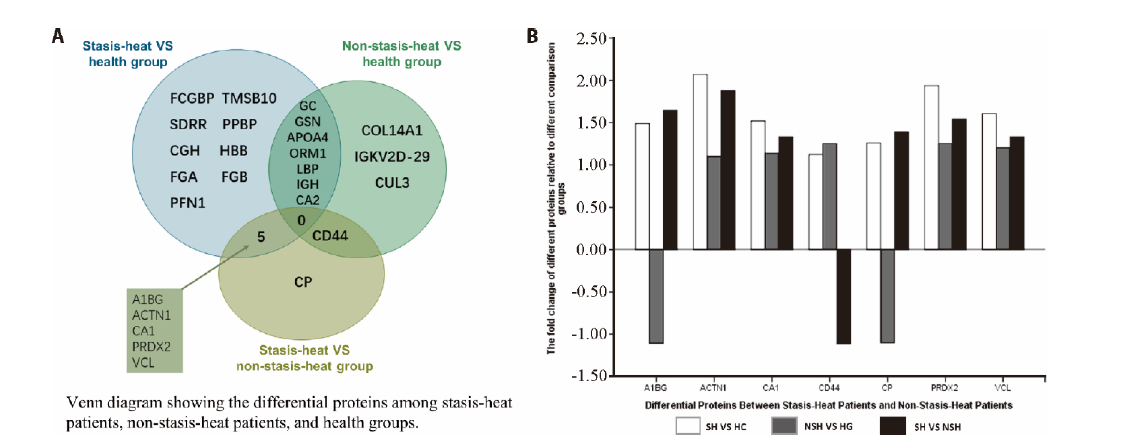
Figure 2 Comparison of differential proteins among stasis-heat patients, non-stasis-heat patients, and health groups A: venn diagram showing the differential proteins among the three groups; B: bar graph showing the expression levels of proteins between stasis-heat and non-stasis-heat group. A1BG: Alpha-1-B glycoprotein; CP: copper-protein; ACTN1: alpha 1; CA1: carbonic anhydrase I; PRDX2: peroxiredoxin 2; VCL: vinculin; SH: stasis-heat; NSH: non-stasis-heat; HC: health control.
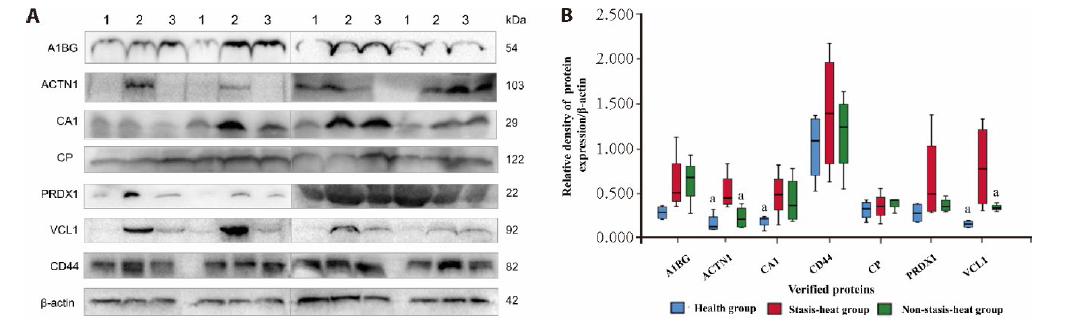
Figure 3 Western blotting analysis of A1BG, ACTN1, CA1, CP, PRDX1, VCL1, and CD44 from healthy controls, stasis-heat group, and non-stasis-heat group A: Western blotting analysis of the seven proteins; B: the intensity of each band was measured using imaging analysis. aP ≤ 0.05, comparison of non-stasis-heat group and healthy group with stasis-heat group separately. A1BG: Alpha-1-B glycoprotein; CP: copper-protein; ACTN1: alpha 1; CA1: carbonic anhydrase I; PRDX2: peroxiredoxin 2; VCL: vinculin.
| 1 |
Zhou M, Wang H, Zeng X, et al. Mortality, morbidity, and risk factors in china and its provinces, 1990-2017: a systematic analysis for the global burden of disease study 2017. Lancet 2019; 394: 1145-58.
DOI URL |
| 2 | Wang Y, Li Z, Gu H, et al. Report on stroke prevention and tre-atment in China. Zhong Guo Zu Zhong Za Zhi 2020; 15: 1037-43. |
| 3 |
Ma H, Campbell BCV, Parsons MW, et al. Thrombolysis guided by perfusion imaging up to 9 hours after onset of stroke. N Engl J Med 2019; 380: 1795-803.
DOI URL |
| 4 |
O'Collins VE, Macleod MR, Donnan GA, Horky LL, van der Worp BH, Howells DW. 1026 experimental treatments in acute stroke. Ann Neurol 2006; 59: 467-77.
DOI PMID |
| 5 | Liu D, Liang J, Kang X, Ma S, Ding M. Molecule prescription: New generative point of science of medicine. Zi Ran Za Zhi 2006; 2006: 337-40. |
| 6 |
Casas AI, Hassan AA, Larsen SJ, et al. From single drug targets to synergistic network pharmacology in ischemic stroke. Proc Natl Acad Sci USA 2019; 116: 7129-36.
DOI URL |
| 7 |
Jiang C, Yang X, Dong J, Li G. Systematic review and Meta-analysis of randomized controlled trials of liangxue tongyu formula on patients with acute intracerebral hemorrhage. Front Pharmacol 2020; 11: 437.
DOI PMID |
| 8 |
Li X, Huang X, Tang Y, et al. Assessing the pharmacological and therapeutic efficacy of traditional chinese medicine liangxue tongyu prescription for intracerebral hemorrhagic stroke in neurological disease models. Front Pharmacol 2018; 9: 1169.
DOI URL |
| 9 |
Guo Q, Yang S, Yang D, et al. Differential mrna expression combined with network pharmacology reveals network effects of liangxue tongyu prescription for acute intracerebral hemorrhagic rats. J Ethnopharmacol 2020; 246: 112231.
DOI URL |
| 10 | Chen Y, Dong J, Yang D, et al. Synergistic network pharmacology for traditional chinese medicine liangxue tongyu formula in acute intracerebral hemorrhagic stroke. Neural Plast 2021; 2021: 8874296. |
| 11 | Huang X, Li GC, Yin L, Zhang ZH, Liang YX, Chen HB. The effective parts of Liangxue Tongyu prescription on cooling-blood and activating-blood and analysis of chemical constituents by hplc-ms and gc-ms. Yao Xue Xue Bao 2015; 50: 86-93. |
| 12 | Guo W, Zhang L, Wu M, et al. Liangxue Tongyu Fang for treating acuter-phase cerebral hemorrhage in 168 cases. Beijing Zhong Yi Yao Da Xue Xue Bao. 2012; 35: 603-6+19. |
| 13 | Tian T, Li G, Guo W. Research of biomarkers in the Yu-Re pathogenic unit of acute cerebral hemorrhage. Shi Zhen Guo Yi Guo Yao 2016; 27: 183-6. |
| 14 | Tian T, Guo W, Li G. Research on clinical manifestation of heat stagnation pathogenesis in acute cerebral hemorrhage. Zhong Yi Za Zhi 2016; 57: 838-42. |
| 15 | Xu X, Chen X, Zhang J, Xu B. Study on the accuracy and reliability of the abc/2 formula for volume assessment of intracerebral he- matoma. Zhong Guo Shen Jing Jing Shen Ji Bing Za Zhi 2015; 41: 87-91. |
| 16 | Li G, Zhou X, Wang J, et al. Research on measuring scale for pathogen unit of hemorrhagic stroke due to heat-stasis. Nanjing Zhong Yi Yao Da Xue Xue Bao 2015; 31: 428-32. |
| 17 |
Li GC, Zhang L, Yu M, et al. Identification of novel biomarker and therapeutic target candidates for acute intracerebral hemorrhage by quantitative plasma proteomics. Clin Proteomics 2017; 14: 14.
DOI URL |
| 18 | Mi H, Muruganujan A, Thomas PD. Panther in 2013: Modeling the evolution of gene function, and other gene attributes, in the context of phylogenetic trees. Nucleic Acids Res 2013; 41: 377-86. |
| 19 | Zhou Z. Research on the syndrome and treatment of intracerebral hemorrhage (haemostasis and heat syndrome). Zhong Yi Yao Xue Kan 2002; 2002: 709-11+23. |
| 20 | Zhou Z. Theory of stasis-heat. Nanjing Zhong Yi Yao Da Xue Xue Bao 2006; 2006: 273-6+331. |
| 21 | Jiang B, Tian L, Xu L. Effects of Liangxue Tongyu on hematological system and brain injury after cerebral ischemia in rats. Zhong Guo Lao Nian Xue Za Zhi 2015; 35: 134-6. |
| 22 |
He C, Huang J, Wang W, et al. Effects of Liangxue Tongyu formula on brain edema and expressions of matrix metalloprooteinase-9 and tissue inhibitor of metalloproteinase-1 in rats with intracerebral hemorrhage. Zhong Xi Yi Jie He Xue Bao 2010; 8: 347-51.
DOI URL |
| 23 |
Schwarz S, Hafner K, Aschoff A, Schwab S. Incidence and prognostic significance of fever following intracerebral hemorrhage. Neurology 2000; 54: 354-61.
PMID |
| 24 |
Rosell A, Cuadrado E, Ortega-Aznar A, Hernandez-Guillamon M, Lo EH, Montaner J. Mmp-9-positive neutrophil infiltration is associated to blood-brain barrier breakdown and basal lamina type iv collagen degradation during hemorrhagic transformation after human ischemic stroke. Stroke 2008; 39: 1121-6.
DOI PMID |
| 25 |
Cao G, Savani RC, Fehrenbach M, et al. Involvement of endothelial cd44 during in vivo angiogenesis. Am J Pathol 2006; 169: 325-36.
DOI URL |
| 26 |
Winder SJ, Ayscough KR. Actin-binding proteins. J Cell Sci 2005; 118: 651-4.
DOI URL |
| 27 |
Bois PR, O'Hara BP, Nietlispach D, Kirkpatrick J, Izard T. The vinculin binding sites of talin and alpha-actinin are sufficient to activate vinculin. J Biol Chem 2006; 281: 7228-36.
DOI URL |
| 28 |
Shah DI, Singh M. Involvement of rho-kinase in experimental vascular endothelial dysfunction. Mol Cell Biochem 2006; 283: 191-9.
DOI URL |
| 29 |
Liu X, Lu D, Bowser R, Liu J. Expression of carbonic anhydrase i in motor neurons and alterations in als. Int J Mol Sci 2016; 17: 1820.
DOI URL |
| 30 |
Yang G, Hu R, Zhang C, et al. A combination of serum iron, ferritin and transferrin predicts outcome in patients with intracerebral hemorrhage. Sci Rep 2016; 6: 21970.
DOI URL |
| 31 |
Liu H, Hua Y, Keep RF, Xi G. Brain ceruloplasmin expression after experimental intracerebral hemorrhage and protection against iron-induced brain injury. Transl Stroke Res 2019; 10: 112-9.
DOI URL |
| 32 |
Tian M, Cui YZ, Song GH, et al. Proteomic analysis identifies MMP-9, DJ-1 and A1BG as overexpressed proteins in pancreatic juice from pancreatic ductal adenocarcinoma patients. BMC Cancer 2008; 8: 241.
DOI PMID |
| 33 | Bai S, Liu S, Guo X, et al. Proteome analysis of biomarkers in the cerebrospinal fluid of neuromyelitis optica patients. Mol Vis 2009; 15: 1638-48. |
| 34 |
Sevilla L, Zaldumbide A, Pognonec P, Boulukos KE. Transcriptional regulation of the Bcl-X gene encoding the anti-apoptotic Bcl-XL protein by ets, rel/nfkappab, stat and ap1 transcription factor families. Histol Histopathol 2001; 16: 595-601.
DOI PMID |
| 35 |
Xi S, Gooding WE, Grandis JR. In vivo antitumor efficacy of STAT3 blockade using a transcription factor decoy approach: implications for cancer therapy. Oncogene 2005; 24: 970-9.
DOI URL |
| 36 |
Liu J, Su G, Gao J, Tian Y, Liu X, Zhang Z. Effects of peroxiredoxin 2 in neurological disorders: a review of its molecular mechanisms. Neurochem Res 2020; 45: 720-30.
DOI URL |
| 37 |
Frantz S, Kobzik L, Kim YD, et al. Toll4 (tlr4) expression in cardiac myocytes in normal and failing myocardium. J Clin Invest 1999; 104: 271-80.
PMID |
| 38 | Zhao F, Huang Y, Li G. Influence of Liangxue Tongyu Fang on proliferation, secretion and related signaling pathway proteins expression in vec. Nanjing Zhong Yi Yao Da Xue Xue Bao 2018; 34: 510-2. |
| 39 |
Norata GD, Ongari M, Uboldi P, Pellegatta F, Catapano AL. Liver x receptor and retinoic x receptor agonists modulate the expression of genes involved in lipid metabolism in human endothelial cells. Int J Mol Med 2005; 16: 717-22.
PMID |
| 40 | Gruys E, Toussaint MJ, Niewold TA, Koopmans SJ. Acute phase reaction and acute phase proteins. J Zhejiang Univ Sci B 2005; 6: 1045-56. |
| 41 |
Hajjar DP, Pomerantz KB. Signal transduction in atherosclerosis: Integration of cytokines and the eicosanoid network. FASEB J 1992; 6: 2933-41.
PMID |
| 42 |
Schmidt A, Hall MN. Signaling to the actin cytoskeleton. Annu Rev Cell Dev Biol 1998; 14: 305-38.
PMID |
| 43 | Carpenter CL. Actin cytoskeleton and cell signaling. Crit Care Med 2000; 4 Suppl(28):N94-9. |
| 44 |
Suzuki M, Ogawa A, Sakurai Y, et al. Thrombin activity in cerebrospinal fluid after subarachnoid hemorrhage. Stroke 1992; 23: 1181-2.
PMID |
| 45 |
Sorimachi H, Ono Y. Regulation and physiological roles of the calpain system in muscular disorders. Cardiovasc Res 2012; 96: 11-22.
DOI PMID |
| 46 |
Nishimura T, Takeichi M. Remodeling of the adherens junctions during morphogenesis. Curr Top Dev Biol 2009; 89: 33-54.
DOI PMID |
| 47 |
Lopez-Colome AM, Lee-Rivera I, Benavides-Hidalgo R, Lopez E. Paxillin: a crossroad in pathological cell migration. J Hematol Oncol 2017; 10: 50.
DOI URL |
| [1] | ZHANG Di, WEI Muyun, CHEN Luan, WU Hao, WANG Ting, ZHANG Zhiruo, ZHANG Ying, YU Juan, HUANG Jinming, ZHU Jinhang, QIN Shengying. Drug response biomarkers of Pien Tze Huang (片仔癀) treatment for hepatic fibrosis induced by carbon tetrachloride [J]. Journal of Traditional Chinese Medicine, 2022, 42(4): 530-538. |
| [2] | Zeng Chuang, Bai Xuejing, Qin Heping, Wang Hong, Rong Xiaofeng, Yan Jin. Effect of adjuvant therapy with electroacupuncture on bone turnover markers and interleukin 17 in patients with rheumatoid arthritis [J]. Journal of Traditional Chinese Medicine, 2019, 39(04): 582-586. |
| [3] | Li Jiun-Yi, Chang Ting-Chen, Chang Nen-Chung, Thanasekaran Jayakumar, Chang Chao-Chien. Anti-embolic effect of Taorenchengqi Tang in rats with embolic stroke induced by occluding middle cerebral artery [J]. Journal of Traditional Chinese Medicine, 2017, 37(03): 326-332. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

