Journal of Traditional Chinese Medicine ›› 2022, Vol. 42 ›› Issue (4): 641-651.DOI: 10.19852/j.cnki.jtcm.2022.04.007
• Reviews • Previous Articles Next Articles
Herbal anthelmintic agents: a narrative review
- Department of Pharmaceutical Sciences and Natural Products, Central University of Punjab, Ghudda, Bathinda, Punjab 151401, India
-
Received:2021-10-16Accepted:2022-01-05Online:2022-08-15Published:2022-07-12 -
Contact:Pradeep Kumar -
About author:Pradeep Kumar, Department of Pharmaceutical Sciences and Natural Products, Central University of Punjab, Ghudda, Bathinda, Punjab 151401, India. pradeepyadav27@gmail.com, Telephone: 9813774553
Cite this article
Manjusa Adak, Pradeep Kumar. Herbal anthelmintic agents: a narrative review[J]. Journal of Traditional Chinese Medicine, 2022, 42(4): 641-651.
share this article
Table 1 Diseases caused by helminths
| Type of Helminths | Species | Diseases |
|---|---|---|
| Cestodes (Tapeworm) | Coenurus cerebralis - In sheep, rabbits, and rodents, - Humans become an intermediate host after ingesting food contaminated. | Coenurosis - Involvement of CNS. -Cysts are developed into ventricles, and sometimes within the parenchyma of the brain, spinal cord. |
| Diphyllobothrium latum - The fish tapeworm, infect humans who ingest raw and pickled freshwater fish. - Compete with hosts for certain vitamins and related substances particularly for vitamin B12 and split vitamin B12 intrinsic factor complex. | Diphyllobothriasis - Low concentration of vitamin B12 - Systemic and neurologic symptoms, including pallor, glossitis, loss of tongue papillae, numbness, paresthesias of feet and hand, depression, loss of vibratory sensation. - Optic neuropathy. | |
| Spirometra species - Cats and dogs (definitive host). - Humans are accidental hosts, acquire infection by drinking contaminated water. - Penetrate intestinal wall and migrate to brain and other tissues and grow to full size. | Sparganosis - Slow-growing, tender, migratory, subcutaneous nodules develop for several weeks or years. - Symptoms like fever, chill, edema, and peripheral eosinophilia. - In rare case, helminths travel to the eye and brain. | |
| Taenia solium - Tapeworms, migrate through the mucosa and enter in the CNS, eyes, and striated muscle. | Cysticercosis - Affects CNS, causes fever, headaches during the larval stage, progressive muscle weakness. | |
| Nematodes | Angiostrongylus cantonensis - Infect mollusks or fish. - In humans, it affects the upper respiratory tract, CNS | Angiostrongyliasis - Characterized by eosinophilic meningitis - Symptoms are rash, pruritus, abdominal pain, headache, stiff neck, vomiting, etc. |
| Gnathostoma spinigerum - Parasitizes the stomachs of cats and dogs. - Migrate through human tissue by ingestion of cooked animal flesh, drinking contaminated water | Gnathostomiasis - Mainly CNS can be affected, the spinal cord is affected initially. - Patients experience nausea, vomiting, upper abdominal pain, urticaria, pruritus, etc. | |
| Loa loa - Group of nematodes, also called filaria. - White, threadlike worms transmitted to humans by biting tabanid flies. | Loiasis - Infections are usually asymptomatic. - To treat loasis, one should consult an expert on tropical infectious diseases. | |
| Onchocerca volvulus - Similar to Loa loa. - Transmitted to humans by female black flies. | Onchocerciasis - Granulomatous reaction followed by fibrosis. - Skin lesions, characterized by erythematous, pruritis, rash, etc. | |
| Strongyloides stercoralis - Small nematodes, can parasitize the small bowel of humans. - This female worm hatch in the intestinal duodenum and jejunum and pass into feces. | Strongyloidiasis - Infection is caused in humans by skin contact with the contaminated soil. - Intestinal parasitism | |
| Toxocara canis and cati - Infects dogs and related mammals. - Eggs hatch into the small intestine of the host and larvae migrate into lungs, and trachea. | Toxocariasis - Human toxocariasis occurs accidentally by ingesting the faeces of infected dogs. - Conditions may develop to the eye, CNS, producing nonspecific systemic manifestations. | |
| Trichinella spiralis - Human infection occurs by eating raw and undercooked pork, bear, wild boar. - After eating, the larvae migrate to the stomach, then to the small intestine. | Trichinosis - Characterized by abdominal pain, vomiting, and diarrhea followed by fever, headache, lethargy, and severe muscle pain, weakness and tenderness. | |
| Paragonimus spices - Lung flukes, humans and other mammals are the final host, first intermediate host are snails, second intermediate host crustaceans. - Penetrating intestinal wall, entering into peritoneal cavity then migrating through diaphragm go to lungs. Schistosoma spices - Humans and other mammals are the final host, first intermediate host are snails, second intermediate host are crustaceans. - Found in intramedullary granuloma, spinal cord, CNS | Paragonimiasis - Most cases mild and asymptomatic. - Brownish sputum with cough and intermittent hemoptysis. - Chronic bronchitis. Schistosomiasis - Involvement of CNS. - Chronic schistosomiasis is more common than acute. - Acute schistosomiasis is caused by an immunological response to helminths. Chronic schistosomiasis is an inflammatory response to eggs. |
Table 1 Diseases caused by helminths
| Type of Helminths | Species | Diseases |
|---|---|---|
| Cestodes (Tapeworm) | Coenurus cerebralis - In sheep, rabbits, and rodents, - Humans become an intermediate host after ingesting food contaminated. | Coenurosis - Involvement of CNS. -Cysts are developed into ventricles, and sometimes within the parenchyma of the brain, spinal cord. |
| Diphyllobothrium latum - The fish tapeworm, infect humans who ingest raw and pickled freshwater fish. - Compete with hosts for certain vitamins and related substances particularly for vitamin B12 and split vitamin B12 intrinsic factor complex. | Diphyllobothriasis - Low concentration of vitamin B12 - Systemic and neurologic symptoms, including pallor, glossitis, loss of tongue papillae, numbness, paresthesias of feet and hand, depression, loss of vibratory sensation. - Optic neuropathy. | |
| Spirometra species - Cats and dogs (definitive host). - Humans are accidental hosts, acquire infection by drinking contaminated water. - Penetrate intestinal wall and migrate to brain and other tissues and grow to full size. | Sparganosis - Slow-growing, tender, migratory, subcutaneous nodules develop for several weeks or years. - Symptoms like fever, chill, edema, and peripheral eosinophilia. - In rare case, helminths travel to the eye and brain. | |
| Taenia solium - Tapeworms, migrate through the mucosa and enter in the CNS, eyes, and striated muscle. | Cysticercosis - Affects CNS, causes fever, headaches during the larval stage, progressive muscle weakness. | |
| Nematodes | Angiostrongylus cantonensis - Infect mollusks or fish. - In humans, it affects the upper respiratory tract, CNS | Angiostrongyliasis - Characterized by eosinophilic meningitis - Symptoms are rash, pruritus, abdominal pain, headache, stiff neck, vomiting, etc. |
| Gnathostoma spinigerum - Parasitizes the stomachs of cats and dogs. - Migrate through human tissue by ingestion of cooked animal flesh, drinking contaminated water | Gnathostomiasis - Mainly CNS can be affected, the spinal cord is affected initially. - Patients experience nausea, vomiting, upper abdominal pain, urticaria, pruritus, etc. | |
| Loa loa - Group of nematodes, also called filaria. - White, threadlike worms transmitted to humans by biting tabanid flies. | Loiasis - Infections are usually asymptomatic. - To treat loasis, one should consult an expert on tropical infectious diseases. | |
| Onchocerca volvulus - Similar to Loa loa. - Transmitted to humans by female black flies. | Onchocerciasis - Granulomatous reaction followed by fibrosis. - Skin lesions, characterized by erythematous, pruritis, rash, etc. | |
| Strongyloides stercoralis - Small nematodes, can parasitize the small bowel of humans. - This female worm hatch in the intestinal duodenum and jejunum and pass into feces. | Strongyloidiasis - Infection is caused in humans by skin contact with the contaminated soil. - Intestinal parasitism | |
| Toxocara canis and cati - Infects dogs and related mammals. - Eggs hatch into the small intestine of the host and larvae migrate into lungs, and trachea. | Toxocariasis - Human toxocariasis occurs accidentally by ingesting the faeces of infected dogs. - Conditions may develop to the eye, CNS, producing nonspecific systemic manifestations. | |
| Trichinella spiralis - Human infection occurs by eating raw and undercooked pork, bear, wild boar. - After eating, the larvae migrate to the stomach, then to the small intestine. | Trichinosis - Characterized by abdominal pain, vomiting, and diarrhea followed by fever, headache, lethargy, and severe muscle pain, weakness and tenderness. | |
| Paragonimus spices - Lung flukes, humans and other mammals are the final host, first intermediate host are snails, second intermediate host crustaceans. - Penetrating intestinal wall, entering into peritoneal cavity then migrating through diaphragm go to lungs. Schistosoma spices - Humans and other mammals are the final host, first intermediate host are snails, second intermediate host are crustaceans. - Found in intramedullary granuloma, spinal cord, CNS | Paragonimiasis - Most cases mild and asymptomatic. - Brownish sputum with cough and intermittent hemoptysis. - Chronic bronchitis. Schistosomiasis - Involvement of CNS. - Chronic schistosomiasis is more common than acute. - Acute schistosomiasis is caused by an immunological response to helminths. Chronic schistosomiasis is an inflammatory response to eggs. |
Table 2 Ion-channels as target sites for anthelmintics
| Parasite group | Target site | Example |
|---|---|---|
| Nematodes | Nicotinic acetylcholine receptor agonists | Levamisole |
| Large intestinal nematodes | Gamma-Aminobutyric acid receptor agonists | Piperazine |
| Nematodes and insect parasites | Glutamate-gated chloride channel receptor potentiators | Ivermectin |
| Cestodes and trematodes | Membrane calcium permeability enhancers | Praziquantel |
Table 2 Ion-channels as target sites for anthelmintics
| Parasite group | Target site | Example |
|---|---|---|
| Nematodes | Nicotinic acetylcholine receptor agonists | Levamisole |
| Large intestinal nematodes | Gamma-Aminobutyric acid receptor agonists | Piperazine |
| Nematodes and insect parasites | Glutamate-gated chloride channel receptor potentiators | Ivermectin |
| Cestodes and trematodes | Membrane calcium permeability enhancers | Praziquantel |
Table 3 Target sites for anthelmintics (other than ion-channels)
| Parasite group | Target site | Example |
|---|---|---|
| Nematodes, cestodes and trematodes | β-tubulin binders | Thiabendazole |
| Blood feeders: flukes, Haemonchus contortus, Oestrus ovis | Proton ionophores | Closantel |
| Immature Fasciola | Malate metabolism inhibitors | Diamphenethide |
| Fasciola | Phosphoglycerate kinase and mutase inhibitors | Clorsulon |
| Filaria | Arachidonic acid metabolism inhibitors | Biethylcarbamazine |
Table 3 Target sites for anthelmintics (other than ion-channels)
| Parasite group | Target site | Example |
|---|---|---|
| Nematodes, cestodes and trematodes | β-tubulin binders | Thiabendazole |
| Blood feeders: flukes, Haemonchus contortus, Oestrus ovis | Proton ionophores | Closantel |
| Immature Fasciola | Malate metabolism inhibitors | Diamphenethide |
| Fasciola | Phosphoglycerate kinase and mutase inhibitors | Clorsulon |
| Filaria | Arachidonic acid metabolism inhibitors | Biethylcarbamazine |
Table 4 Plants having anthelmintic activity
| Name | Biological sources | Mechanism of action | Chemical constituents |
|---|---|---|---|
| Myrrh | Commiphora myrrha or Commiphora molmol, Family-Burseraceae | Reduces activities of Alanine transminase (ALT) and Aspartate transminase (AST) | Limonene, Eugenol, a-Pinene, Cadinene, Acetic acid, Formic acid etc. |
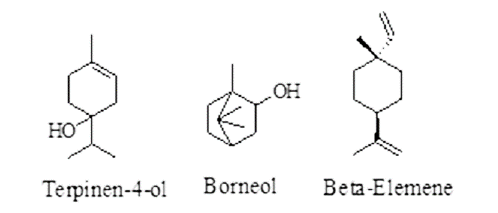
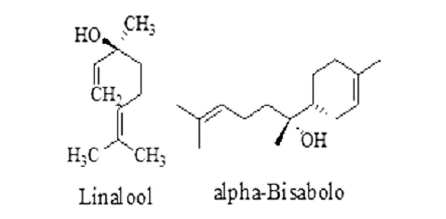
| Tulsi | Ocimum sanctum Linn. Family-Lamiaceae | Causing paralysis of infected parasitic worms or death. | Carvacrol, Caryophyllene, Eugenol, Linalool, Urosolic acid, etc. |
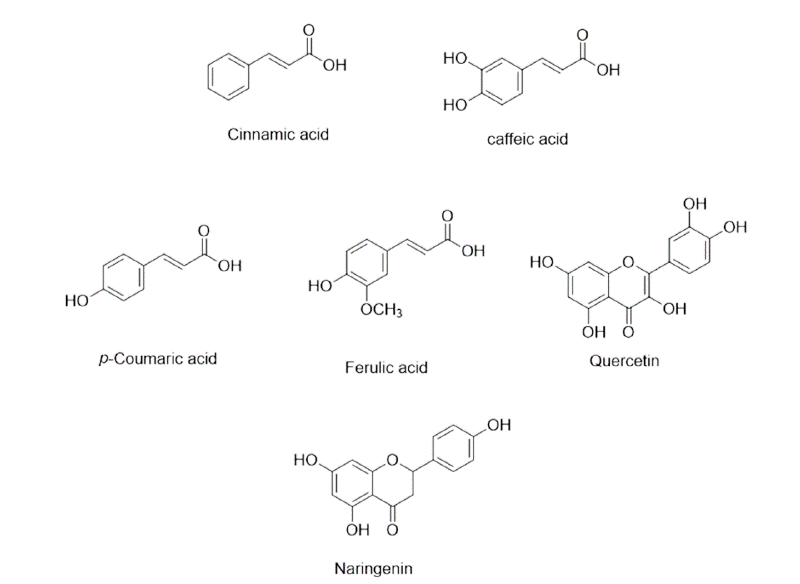
| Chinaberry tree | Melia azedarach, Family- Meliaceae | Reacting with free proteins reduces the nutrients availability, thus larval death occurs due to starvation, or React with glycoproteins in the larval cuticle, causing death. | Spathulenol, Quercetin, Astragalin, 1,7,8-Trihydroxy-2-naphtaldehyde etc. |
| Papaya | Carica papaya, Family-Caricaceae | Killing the parasite worms by eosinophils, attack on structural protein of parasite nematodes. | Papain, Cystatin, Chymopapain, Ascorbic acid, Tocopherol. |
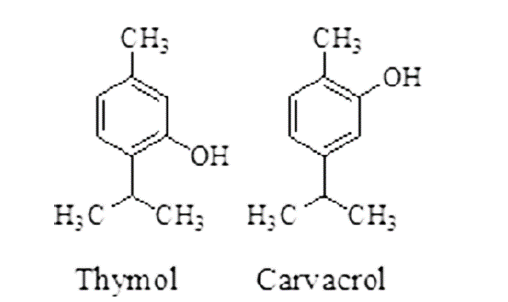
| Black caraway | Nigella sativa, Family-Ranunculaceae | Inhibiting the antioxidant enzymes thus produces a defense mechanism towards the oxidants generated by the parasitic nematode. | Linoleic acid, Oleic acid, Palmitic acid, p-cymene, Carvacrol, Thymol, α-Pinene. |
| Sohphlang | Flemingia vestita, Family-Leguminosae | Causing paralysis of infected parasitic worms or death. | Formononetin, Genistein, Daidzein, Pseudobaptigenin. |
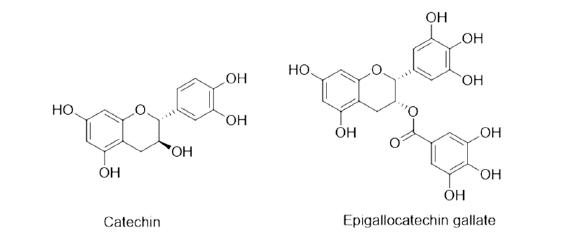
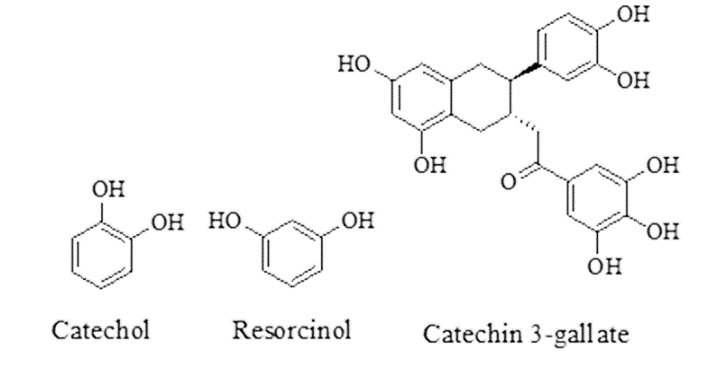
| Walnut | Juglans regia, Family-Juglandaceae | It binds with the free protein of GIT of the host or interferes in energy generation of helminths, causing death of parasites. | Stearic acid, Palmitic acid, alpha Linolenic acid, Oleic acid, Catechin, Tannins. |
| Mimusops | Mimusops elengi, Family-Sapotaceae | Denaturation of proteins, produce defense mechanism, damages reactive oxygen species (ROS) properties. | Ursolic acid, Spinasterol, Taraxerol etc. |
| Pomegranate | Punica granatum Family-Punicaceae. | Inhibit transformation of larvae from egg, produce inflammation of epithelial cells by peroxisome proliferator-activated receptors-γ and δ-dependent mechanisms | Ellagic acid, Cyanidin-3-glucose, Pelargonidin-3-glucose etc. |
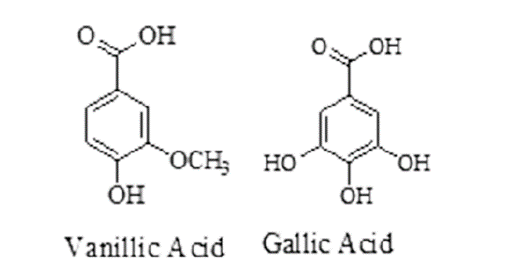
| Embelia | Embelia ribes, Family-Primulaceae | Paralysis of worms and reduces fecal eggs per gram (EPG). | Vanillic acid, Christembine, Cinnamic acid, O-cumaric acid, Embelin. |
| Epazote | Chenopodium ambrosioides, Family-Amaranthaceae. | Paralysis of the parasitic worms. | Limonene, α-Terpinene p-Cymene, Camphor Thymol. |
| Piliostigma | Piliostigma thonningii, Family-Fabaceae. | It stimulates the neuromuscular junction of the parasite mostly and sometimes its effects to the ganglion and cause larval paralysis. | Alepterolic acid, Anticopalic acid, Clovane-2β,9α-diol etc. |
| Asparagus | Asparagus officinalis, Asparagus racemosus, Family-Asparagaceae. | Causing paralysis of infected parasitic worms or death. | Racemosol, Asparagamine, Folic acid. |
| White clover | Trifolium repens, Family-Fabaceae. | Paralysis of worms and reduces EPG. | Rutin, Quercetin, Myricetin Kaempferol. |
| Fig | Ficus insipida, Family-Moraceae. | Causing paralysis of infected parasitic worms or death. | Vomifoliol, Dihydrophaseic acid, Dehydrovomifoliol etc. |
| Squash | Cucurbita maxima, Family-Cucurbitaceae. | Inhibit transformation of larvae from egg, reduces EPG. | Palmitic acid, Oleic acid, Linoleic acid, β-sitosterol. |
| Ajwain | Trachyspermum ammi, Family-Apiaceae or Umbelliferae. | Causing paralysis of infected parasitic worms or death. | Thymol, α-Pinene, α-Terpinene, β-Pinene, γ-terpinene, p-cymene. |
| Cinnamon | Cinnamomum zylanicum, Family - Lauraceae. | Inhibition of the parasitic egg hatching inhibits the fourth stage of larvae motility. | Eugenol, Cinnamic acid Cymene, Cinnamate. |
| Nutmeg | Myristica fragrans, Family -Myristicaceae. | Causing paralysis by inhibiting acetyl cholinesterase. | Myristicin, Eugenol, Safrole, Terpinene, Myristic acid. |
| Elecampane | Inula helenium, Family-Asteraceae. | Inhibitory effects on process of embryo development, paralysis by inhibiting acetylcholinesterase. | Alantolactone, Inulin, Helenin Stearoptene. |
| Clausena anisata | Clausena anisata, Family-Rutaceae. | Causing paralysis by inhibiting acetylcholinesterase. | Coumarins, Linalool, Myrcene, Anethole, Lomonene etc. |
| Zanthoxylum | Zanthoxylum zanthoxyloides, Family-Rutaceae. | Inhibition of the parasitic egg hatching prevents larvae from migrating. | Lomonine, Citronellal, Myrcene, α-pinene. |
| Annona | Annona squamosa, Family-Annonaceae. | Inhibition of the parasitic egg hatching inhibits cell division. | Anonain, OxophoebineIsocorydine, Reticulin. |
| False daisy | Eclipta prostrata, Family-Asteraceae. | Causing paralysis of infected parasitic worms or death. | Quercetin, β-Sitosterol, Luteoloside, Apigenin, Luteolin. |
| Name | Biological sources | Mechanism of action | Chemical constituents |
| Turkey berry | Solanum torvum, Family-Solanaceae. | Causing paralysis of infected parasitic worms, or reduces EPG. | Quercetin, Isoquarecetin Kaempferol, Rutin etc. |
| Myrobalan | Terminalia chebula, Family -Combretaceae. | Interrupts in energy production by binds free protein from GI tract or oxidative phosphorylation. | Arjungenin, Chebulin, Ellagic acid, Chebulic acid, Gallic acid. |
| Vinca | Catharanthus roseus, Family-Apocynaceae | Prevents polymerization of tubulin into microtubules. | Vincristine, Vinblastine Catharanthine etc. |
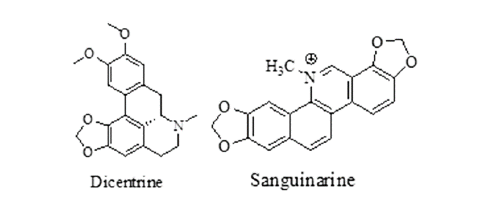
| Celandine | Chelidonium majus, Family-Papaveraceae | Reduce ROS generation, paralysis the parasitic worms. | Chelidonine, Sanguinarine, Caffeic acid, Protopine. |
| Mentha | Mentha cordifolia, Family-Lamiaceae | Causing paralysis of infected parasitic worms or death. | Carvone, Limonene, Menthol. |
| Sainfoin | Onobrychis viciifolia, Family-Fabaceae. | Reduce nematode excretion from GI tract, delay in egg maturation. | Tannin, Rutin, Nicotiflorin. |
| Ashwagandha | Withania somnifera, Family -Solanaceae | Causing paralysis of infected parasitic worms or death. | Withanolides, Anaferine, Sitoindoside. |
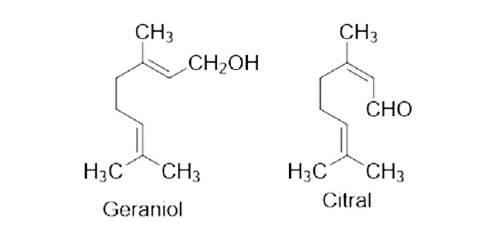
| Coriander | Coriandrum sativum, Family - Apiaceae | Reduce faecal egg count of worm and also inhibit the egg hatching process. | Linalool, Camphor, Geraniol, Coumarins, Linoleic acid. |
Table 4 Plants having anthelmintic activity
| Name | Biological sources | Mechanism of action | Chemical constituents |
|---|---|---|---|
| Myrrh | Commiphora myrrha or Commiphora molmol, Family-Burseraceae | Reduces activities of Alanine transminase (ALT) and Aspartate transminase (AST) | Limonene, Eugenol, a-Pinene, Cadinene, Acetic acid, Formic acid etc. |
| Tulsi | Ocimum sanctum Linn. Family-Lamiaceae | Causing paralysis of infected parasitic worms or death. | Carvacrol, Caryophyllene, Eugenol, Linalool, Urosolic acid, etc. |
| Chinaberry tree | Melia azedarach, Family- Meliaceae | Reacting with free proteins reduces the nutrients availability, thus larval death occurs due to starvation, or React with glycoproteins in the larval cuticle, causing death. | Spathulenol, Quercetin, Astragalin, 1,7,8-Trihydroxy-2-naphtaldehyde etc. |
| Papaya | Carica papaya, Family-Caricaceae | Killing the parasite worms by eosinophils, attack on structural protein of parasite nematodes. | Papain, Cystatin, Chymopapain, Ascorbic acid, Tocopherol. |
| Black caraway | Nigella sativa, Family-Ranunculaceae | Inhibiting the antioxidant enzymes thus produces a defense mechanism towards the oxidants generated by the parasitic nematode. | Linoleic acid, Oleic acid, Palmitic acid, p-cymene, Carvacrol, Thymol, α-Pinene. |
| Sohphlang | Flemingia vestita, Family-Leguminosae | Causing paralysis of infected parasitic worms or death. | Formononetin, Genistein, Daidzein, Pseudobaptigenin. |
| Walnut | Juglans regia, Family-Juglandaceae | It binds with the free protein of GIT of the host or interferes in energy generation of helminths, causing death of parasites. | Stearic acid, Palmitic acid, alpha Linolenic acid, Oleic acid, Catechin, Tannins. |
| Mimusops | Mimusops elengi, Family-Sapotaceae | Denaturation of proteins, produce defense mechanism, damages reactive oxygen species (ROS) properties. | Ursolic acid, Spinasterol, Taraxerol etc. |
| Pomegranate | Punica granatum Family-Punicaceae. | Inhibit transformation of larvae from egg, produce inflammation of epithelial cells by peroxisome proliferator-activated receptors-γ and δ-dependent mechanisms | Ellagic acid, Cyanidin-3-glucose, Pelargonidin-3-glucose etc. |
| Embelia | Embelia ribes, Family-Primulaceae | Paralysis of worms and reduces fecal eggs per gram (EPG). | Vanillic acid, Christembine, Cinnamic acid, O-cumaric acid, Embelin. |
| Epazote | Chenopodium ambrosioides, Family-Amaranthaceae. | Paralysis of the parasitic worms. | Limonene, α-Terpinene p-Cymene, Camphor Thymol. |
| Piliostigma | Piliostigma thonningii, Family-Fabaceae. | It stimulates the neuromuscular junction of the parasite mostly and sometimes its effects to the ganglion and cause larval paralysis. | Alepterolic acid, Anticopalic acid, Clovane-2β,9α-diol etc. |
| Asparagus | Asparagus officinalis, Asparagus racemosus, Family-Asparagaceae. | Causing paralysis of infected parasitic worms or death. | Racemosol, Asparagamine, Folic acid. |
| White clover | Trifolium repens, Family-Fabaceae. | Paralysis of worms and reduces EPG. | Rutin, Quercetin, Myricetin Kaempferol. |
| Fig | Ficus insipida, Family-Moraceae. | Causing paralysis of infected parasitic worms or death. | Vomifoliol, Dihydrophaseic acid, Dehydrovomifoliol etc. |
| Squash | Cucurbita maxima, Family-Cucurbitaceae. | Inhibit transformation of larvae from egg, reduces EPG. | Palmitic acid, Oleic acid, Linoleic acid, β-sitosterol. |
| Ajwain | Trachyspermum ammi, Family-Apiaceae or Umbelliferae. | Causing paralysis of infected parasitic worms or death. | Thymol, α-Pinene, α-Terpinene, β-Pinene, γ-terpinene, p-cymene. |
| Cinnamon | Cinnamomum zylanicum, Family - Lauraceae. | Inhibition of the parasitic egg hatching inhibits the fourth stage of larvae motility. | Eugenol, Cinnamic acid Cymene, Cinnamate. |
| Nutmeg | Myristica fragrans, Family -Myristicaceae. | Causing paralysis by inhibiting acetyl cholinesterase. | Myristicin, Eugenol, Safrole, Terpinene, Myristic acid. |
| Elecampane | Inula helenium, Family-Asteraceae. | Inhibitory effects on process of embryo development, paralysis by inhibiting acetylcholinesterase. | Alantolactone, Inulin, Helenin Stearoptene. |
| Clausena anisata | Clausena anisata, Family-Rutaceae. | Causing paralysis by inhibiting acetylcholinesterase. | Coumarins, Linalool, Myrcene, Anethole, Lomonene etc. |
| Zanthoxylum | Zanthoxylum zanthoxyloides, Family-Rutaceae. | Inhibition of the parasitic egg hatching prevents larvae from migrating. | Lomonine, Citronellal, Myrcene, α-pinene. |
| Annona | Annona squamosa, Family-Annonaceae. | Inhibition of the parasitic egg hatching inhibits cell division. | Anonain, OxophoebineIsocorydine, Reticulin. |
| False daisy | Eclipta prostrata, Family-Asteraceae. | Causing paralysis of infected parasitic worms or death. | Quercetin, β-Sitosterol, Luteoloside, Apigenin, Luteolin. |
| Name | Biological sources | Mechanism of action | Chemical constituents |
| Turkey berry | Solanum torvum, Family-Solanaceae. | Causing paralysis of infected parasitic worms, or reduces EPG. | Quercetin, Isoquarecetin Kaempferol, Rutin etc. |
| Myrobalan | Terminalia chebula, Family -Combretaceae. | Interrupts in energy production by binds free protein from GI tract or oxidative phosphorylation. | Arjungenin, Chebulin, Ellagic acid, Chebulic acid, Gallic acid. |
| Vinca | Catharanthus roseus, Family-Apocynaceae | Prevents polymerization of tubulin into microtubules. | Vincristine, Vinblastine Catharanthine etc. |
| Celandine | Chelidonium majus, Family-Papaveraceae | Reduce ROS generation, paralysis the parasitic worms. | Chelidonine, Sanguinarine, Caffeic acid, Protopine. |
| Mentha | Mentha cordifolia, Family-Lamiaceae | Causing paralysis of infected parasitic worms or death. | Carvone, Limonene, Menthol. |
| Sainfoin | Onobrychis viciifolia, Family-Fabaceae. | Reduce nematode excretion from GI tract, delay in egg maturation. | Tannin, Rutin, Nicotiflorin. |
| Ashwagandha | Withania somnifera, Family -Solanaceae | Causing paralysis of infected parasitic worms or death. | Withanolides, Anaferine, Sitoindoside. |
| Coriander | Coriandrum sativum, Family - Apiaceae | Reduce faecal egg count of worm and also inhibit the egg hatching process. | Linalool, Camphor, Geraniol, Coumarins, Linoleic acid. |
| 1 | Fenwick A. The global burden of neglected tropical diseases. J Public Health 2012; 126: 233-6. |
| 2 |
Hotez PJ, Bottazzi ME, Strych U. New vaccines for the world's poorest people. Annu Rev Med 2016; 67: 405-17.
DOI URL |
| 3 | Knox M, Besier R, Le LJ. Novel approaches to the control of helminth parasites of livestock. Foreword. Vet Parasitol 2012; 186: 1-1. |
| 4 |
Stepek G, Behnke JM, Buttle DJ, et al. Natural plant cysteine proteinases as anthelmintics? Trends Parasitol 2004; 20: 322-7.
DOI URL |
| 5 |
Kappus KD, Lundgren Jr RG, Juranek DD, et al. Intestinal parasitism in the United States: Update on a continuing problem. Am J Trop Med Hyg 1994; 50: 705-13.
PMID |
| 6 |
Sharpe C, Thornton DJ, Grencis RK. A sticky end for gastrointestinal helminths; the role of the mucus barrier. Parasite Immunol 2018; 40: e12517.
DOI URL |
| 7 | Preston S, Jiao Y, Baell JB, et al. Screening of the ‘Open Scaffolds’ collection from compounds Australia identifies a new chemical entity with anthelmintic activities against different developmental stages of the barber's pole worm and other parasitic nematodes. Int J Parasitol 2017; 7: 286-94. |
| 8 |
Liu M, Panda SK, Luyten W. Plant-based natural products for the discovery and development of novel anthelmintics against nematodes. Biomolecules 2020; 10: 426.
DOI URL |
| 9 |
Hu Y, Georghiou SB, Kelleher AJ, et al. Bacillus thuringiensis Cry5B protein is highly efficacious as a single-dose therapy against an intestinal roundworm infection in mice. PLoS Negl Trop Dis 2010; 4: e614.
DOI URL |
| 10 | Bogitsh BJ, Carter CE, Oeltmann TN. Intestinal Nematodes. In: Human parasitology. 5th ed. Massachusetts: Academic Press, 2019: 277-311. |
| 11 |
Hotez PJ. Neglected parasitic infections and poverty in the United States. PLoS Negl Trop Dis 2014; 8: e3012.
DOI URL |
| 12 |
Brooker S, Kabatereine N, Gyapong J, et al. Rapid mapping of schistosomiasis and other neglected tropical diseases in the context of integrated control programmes in Africa. Parasitol 2009; 136: 1707-18.
DOI URL |
| 13 |
Geary TG, Chibale K, Abegaz B, et al. A new approach for anthelmintic discovery for humans. Trends Parasitol 2012; 28: 176-81.
DOI URL |
| 14 | Minciullo P, Cascio A, David A, et al. Anaphylaxis caused by helminths: review of the literature. Eur Rev Med Pharmacol Sci 2012; 16: 1513-8. |
| 15 | van Riet E, Hartgers FC, Yazdanbakhsh M. Chronic helminth infections induce immunomodulation: Consequences and mechanisms. J Immunobiol 2007; 212: 475-90. |
| 16 |
Kaewkes S. Taxonomy and biology of liver flukes. Acta Tropica 2003; 88: 177-86.
DOI URL |
| 17 |
Tsai IJ, Zarowiecki M, Holroyd N, et al. The genomes of four tapeworm species reveal adaptations to parasitism. Nature 2013; 496: 57-63.
DOI URL |
| 18 |
Traversa D. Pet roundworms and hookworms: a continuing need for global worming. Parasit Vectors 2012; 5: 91.
DOI PMID |
| 19 |
Bethony J, Brooker S, Albonico M, et al. Soil-transmitted helminth infections: ascariasis, trichuriasis, and hookworm. Lancet 2006; 367: 1521-32.
DOI URL |
| 20 |
Sabattani S, Marliani AF, Roncaroli F, et al. Cerebral coenurosis: case illustration. J Neurosurg 2004; 100: 964.
PMID |
| 21 | Marcial-Rojas RA. Pathology of protozoal and helminthic diseases, with clinical correlation. Baltimore: Williams & Wilkins Co. Edinburgh: Churchill Livingstone; 1971: 698-710. |
| 22 |
Ohnishi K, Murata M. Single dose treatment with praziquantel for human diphyllobothrium nihonkaiense infections. Trans R Soc Trop Med Hyg 1993; 87: 482-3.
DOI URL |
| 23 |
Gökçek C, Bayar N, Buharal Z. Total removal of an unruptured orbital hydatid cyst. Can J Ophthalmol 2001; 36: 218-20.
PMID |
| 24 | Demir K, Karsli A, Kaya T, et al. Cerebral hydatid cysts: CT findings. Neuroradiol J 1991; 33: 22-4. |
| 25 | Botterel F, Bourée P. Ocular sparganosis:a case report. J Travel Med 2003; 10: 245-6. |
| 26 |
Pushker N, Bajaj MS, Betharia SM. Orbital and adnexal cysticercosis. Clin Exp Ophthalmol 2002; 30: 322-33.
PMID |
| 27 |
Hughes A, Biggs B. Parasitic worms of the central nervous system: An Australian perspective. Intern Med J 2002; 32: 541-53.
PMID |
| 28 |
Niu M, Duma R. Meningitis due to protozoa and helminths. Infect Dis Clin North Am 1990; 4: 809-41.
DOI URL |
| 29 | Remme J, Boatin B, Boussinesq M. Helminthic diseases:onchocerciasis and loiasis. In: Quah SR, Cockerham WC, editors. International encyclopedia of public health. 2nd ed. DC: Academic Press; 2017: 576-87. |
| 30 |
Marti H, Haji HJ, Savioli L, et al. A comparative trial of a single-dose ivermectin versus three days of albendazole for treatment of Strongyloides stercoralis and other soil-transmitted helminth infections in children. Am J Trop Med Hyg 1996; 55: 477-81.
PMID |
| 31 | Ament CS, Young LH. Ocular manifestations of helminthic infections: Onchocersiasis, cysticercosis, toxocariasis, and diffuse unilateral subacute neuroretinitis. Int Ophthalmol Clin 2006; 46: 1-10. |
| 32 |
Grove DI, Mahmoud A, Warren KS. Eosinophils and resistance to Trichinella spiralis. J Exp Med 1977; 145: 755-9.
DOI PMID |
| 33 |
Katz M. Anthelmintics. Drugs 1977; 13: 124-36.
PMID |
| 34 | De los Reyes-Gavilán CG, Fernández M, Hudson JA, et al. Role of microorganisms present in dairy fermented products in health and disease. BioMed Res Int 2015; |
| 35 | Saltveit ME. Fruit and vegetable phytochemicals: chemistry and human health, synthesis and metabolism of phenolic compounds. 2nd ed. Hoboken: John Wiley & Sons, 2017: 115-24. |
| 36 | Chanda S, Ramachandra T. A review on some Therapeutic aspects of phytochemicals present in medicinal plants. Int J Pharm Life Sci 2019; 10: 6052-8. |
| 37 |
Mukherjee N, Mukherjee S, Saini P, et al. Phenolics and Terpenoids; The promising new search for anthelmintics: A critical review. Mini Rev Med Chem 2016; 16: 1415-41.
PMID |
| 38 |
Qi H, Wang W, Dai J, et al. In vitro anthelmintic activity of Zanthoxylum simulans essential oil against Haemonchus contortus. Vet Parasitol 2015; 211: 223-7.
DOI PMID |
| 39 | Overend W. 9 Glycosides. Carbohydr 2012; 1: 279. |
| 40 | Kaingu F, Kibor A, Waihenya R, et al. Efficacy of Aloe secundiflora crude extracts on Ascaridia galli in vitro. Sustain Agric Res 2013; 2: 49-53. |
| 41 | Hussein RA, El-Anssary AA. Herbal medicine, plants secondary metabolites. In: the key drivers of the pharmacological actions of medicinal plants. 2018; 11-30. |
| 42 |
Güçlü-Üstündağ Ö, Mazza G. Saponins: properties, applications and processing. Crit Rev Food Sci Nutr 2007; 47: 231-58.
PMID |
| 43 | Ali N, Shah SWA, Shah I, et al. Cytotoxic and anthelmintic potential of crude saponins isolated from Achillea Wilhelmsii C. Koch and Teucrium Stocksianum boiss. BMC Compl Alternative Med 2011; 11: 106. |
| 44 |
Cavalcante GS, de Morais SM, Andre WP, et al. Chemical composition and in vitro activity of Calotropis procera (Ait.) latex on Haemonchus contortus. Vet Parasitol 2016; 226: 22-5.
DOI PMID |
| 45 | Ohri P, Pannu SK. Effect of phenolic compounds on nematodes. J Nat Appl Sci 2010; 2: 344-50. |
| 46 |
Symeonidou I, Bonos E, Moustakidis K, et al. Botanicals: a natural approach to control ascaridiosis in poultry. J Hellenic Vet Med Soc 2018; 69: 711-22.
DOI URL |
| 47 |
Del Carmen Acevedo-Ramírez PM, Hallal-Calleros C, Flores-Pérez I, et al. Anthelmintic effect and tissue alterations induced in vitro by hydrolysable tannins on the adult stage of the gastrointestinal nematode Haemonchus contortus. Vet Parasitol 2019; 266: 1-6.
DOI PMID |
| 48 | Badarina I, Putranto HD, Sulistyowati E. In vitro anthelmintic activity of the extract of coffee husk fermented with Pleurotus ostreatus for Ascaridia galli. Anim Prod Sci 2017; 19: 55-60. |
| 49 |
Huang T, Jander G, de Vos M. Non-protein amino acids in plant defense against insect herbivores: representative cases and opportunities for further functional analysis. Phytochemistry 2011; 72: 1531-7.
DOI URL |
| 50 | Swargiary A, Roy B. In vitro anthelmintic efficacy of Alpinia nigra and its bioactive compound, astragalin against Fasciolopsis buski. Int J Pharm Pharm Sci 2015; 7: 30-5. |
| 51 |
Lei J, Leser M, Enan E. Nematicidal activity of two monoterpenoids and SER-2 tyramine receptor of Caenorhabditis elegans. Biochem Pharmacol 2010; 79: 1062-71.
DOI URL |
| 52 |
Vildina JD, Kalmobe J, Djafsia B, et al. Anti-onchocerca and anti-caenorhabditis activity of a hydro-alcoholic extract from the fruits of Acacia nilotica and some proanthocyanidin derivatives. Molecules 2017; 22: 748.
DOI URL |
| 53 |
Ndjonka D, Abladam E, Djafsia B, et al. Anthelmintic activity of phenolic acids from the axlewood tree Anogeissus leiocarpus on the filarial nematode Onchocerca ochengi and drug-resistant strains of the free-living nematode Caenorhabditis elegans. J Helminthol 2014; 88: 481-8.
DOI PMID |
| 54 |
Pereira I, Severino P, Santos AC, et al. Linalool bioactive properties and potential applicability in drug delivery systems. Colloids Surf B Biointerfaces 2018; 171: 566-78.
DOI URL |
| 55 |
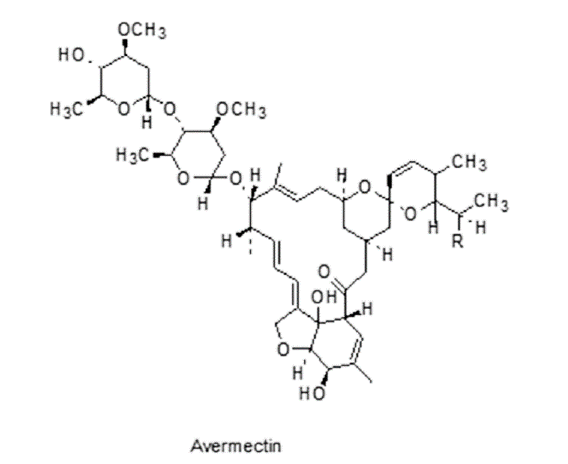
Burg RW, Miller BM, Baker EE, et al. Avermectins, new family of potent anthelmintic agents: producing organism and fermentation. Antimicrob Agents Chemother 1979; 15: 361-7.
DOI PMID |
| 56 |
Yakoot M. A short review of the anthelmintic role of Mirazid. Arq Gastroenterol 2010; 47: 393-4.
DOI URL |
| 57 |
Basyoni MM, El-Sabaa AAA. Therapeutic potential of myrrh and ivermectin against experimental Trichinella spiralis infection in mice. Korean J Parasitol 2013; 51: 297-304.
DOI PMID |
| 58 |
Asha M, Prashanth D, Murali B, et al. Anthelmintic activity of essential oil of Ocimum sanctum and eugenol. Fitoterapia 2001; 72: 669-70.
PMID |
| 59 | Kirtiman S. Comparative study of Withania somnifera and Ocimum sanctum for anthelmintic activity. ISCA J Bio Sci 2012; 1: 74-6. |
| 60 |
Cala A, Chagas A, Oliveira M, et al. In vitro anthelmintic effect of Melia azedarach L. and Trichilia claussenii C. against sheep gastrointestinal nematodes. Exp Parasitol 2012; 130: 98-102.
DOI PMID |
| 61 |
Cala AC, Ferreira JF, Chagas ACS, et al. Anthelmintic activity of Artemisia annua L. extracts in vitro and the effect of an aqueous extract and artemisinin in sheep naturally infected with gastrointestinal nematodes. Parasitol Res 2014; 113: 2345-53.
DOI URL |
| 62 | Kanthal LK, Mondal P, De S, et al. Evaluation of anthelmintic activity of carica papaya latex using Pheritima posthuma. Int J Life Sci Pharma Res 2012; 2: 10-2. |
| 63 | Selvaraju A, Dhanraj S. Phytochemical analysis and anthelmintic potential of Nigella sativa against the trematode, Cotylophoron cotylophorum. J Pharmacogn Phytochem 2019; 8: 3161-6. |
| 64 |
Tandon V, Das B. In vitro testing of anthelmintic efficacy of Flemingia vestita (Fabaceae) on carbohydrate metabolism in Rallietina echinobothrida. Methods 2007; 42: 330-8.
PMID |
| 65 | Kale AA, Gaikwada SA, Kamble G. In vitro anthelmintic activity of stem bark of Juglans regia L. J Chem Pharm Res 2011; 3: 298-302. |
| 66 | Jana GK, Dhanamjayarao M, Vani M. Evaluation of anthelmintic potential of Mimusops elengi Linn (sapotaceae) leaf. J Pharm Res 2010; 3: 2514-5. |
| 67 | Ahmed AH, Ejo M, Feyera T, et al. In vitro anthelmintic activity of crude extracts of artemisia herba-alba and Punica granatum against Haemonchus contortus. J Parasitol 2020; 1-7. |
| 68 |
Ferreira LE, Benincasa BI, Fachin AL, et al. Thymus vulgaris L. essential oil and its main component thymol: anthelmintic effects against Haemonchus contortus from sheep. Vet Parasitol 2016; 228: 70-6.
DOI PMID |
| 69 |
Gundamaraju R. Evaluation of anti-helmintic activity of Ferula foetida “Hing-A natural Indian spice” aqueous extract. Asian Pac J Trop Dis 2013; 3: 189-91.
DOI URL |
| 70 | Sen D, Agnihotri RK, Sharma D, et al. In vitro assays on mangifera indica and embelia ribes against Ascaridia galli of poultry. Himachal J Agric Res 2018; 44: 117-24. |
| 71 |
Hördegen P, Cabaret J, Hertzberg H, et al. In vitro screening of six anthelmintic plant products against larval Haemonchus contortus with a modified methyl-thiazolyl-tetrazolium reduction assay. J Ethnopharmacol 2006; 108: 85-9.
PMID |
| 72 |
Zamilpa A, García-Alanís C, López-Arellano M, et al. In vitro nematicidal effect of Chenopodium ambrosioides and Castela tortuosa n-hexane extracts against Haemonchus contortus (Nematoda) and their anthelmintic effect in gerbils. J Helminthol 2019; 93: 434-9.
DOI PMID |
| 73 | Ali N, Nabi M, Shoaib M, et al. GC/MS analysis, anti-leishmanial and relaxant activity of essential oil of Chenopodium ambrosioides from Malakand region. Pak J Pharm Sci 2021; 34: 577-83. |
| 74 |
Rodrigues JGM, Albuquerque PSV, Nascimento JR, et al. The immunomodulatory activity of Chenopodium ambrosioides reduces the parasite burden and hepatic granulomatous inflammation in Schistosoma mansoni-infection. J Ethnopharmacol 2021; 264: 113287.
DOI URL |
| 75 | Mali RG, Mehta AA. A review on anthelmintic plants. Nat Prod Rad 2008; 7: 466-75. |
| 76 |
Boyko OO, Kabar A, Brygadyrenko V. Nematicidal activity of aqueous tinctures of medicinal plants against larvae of the nematodes Strongyloides papillosus and Haemonchus contortus. Biosyst Divers 2020; 28: 119-23.
DOI URL |
| 77 |
Wang GX, Jiang Dx, Zhou Z, et al. In vivo assessment of anthelmintic efficacy of ginkgolic acids on removal of Ps-eudodactylogyrus in European eel. Aquaculture 2009; 297: 38-43.
DOI URL |
| 78 | Bhavare V, Pokharka R. Comparative in vitro anticestodal activity of some medicinal plants from western India. Pharmacologyonline 2010; 3: 142-5. |
| 79 |
Yadav AK. In vitro anthelmintic assessment of selected phytochemicals against Hymenolepis diminuta, a zoonotic tapeworm. J Parasit Dis 2016; 40: 1082-6.
DOI URL |
| 80 |
Tangpu V, Temjenmongla K, Yadav AK. Anticestodal activity of Trifolium repens extract. Pharm Biol 2005; 42: 656-8.
DOI URL |
| 81 |
Gonzales APPF, Santos GG, Tavares-Dias M. Anthelminthic potential of the Ficus insipida latex on monogeneans of Colossoma macropomum (Serrasalmidae), a medicinal plant from the Amazon. Acta Parasitol 2019; 64: 927-31.
DOI URL |
| 82 | Chand J, Naaz Y, Nainwal P. In vitro Anthelmintic activity of peel extracts of Cucurbita Maxima. Int Res J Pharm 2019; 10: 22-5. |
| 83 |
Lateef M, Iqbal Z, Akhtar M, et al. Preliminary screening of Trachyspermum ammi seed for anthelmintic activity in sheep. Trop Anim Health Prod 2006; 38: 491-6.
PMID |
| 84 | Patil R, Kadam J, Chavan J, et al. Anthelmintic activity of ethanolic bud extract of Syzygium aromaticum against Pheretima posthuma. Weekly Sci Int Res J 2013; 1-5. |
| 85 | Dhanraj KM, Veerakumari L. In vitro effect of Syzygium aromaticum on the motility and acetylcholinesterase of Cotylophoron cotylophorum. Ind J Vet Anim Sci Res 2014; 43: 187-94. |
| 86 |
Cala A, Chagas A, Oliveira M, et al. In vitro anthelmintic effect of Melia azedarach L. and Trichilia claussenii C. against sheep gastrointestinal nematodes. Exp Parasitol 2012; 130: 98-102.
DOI PMID |
| 87 | Kirtiman S. Comparative study of Withania somnifera and Ocimum sanctum for anthelmintic activity. ISCA J Bio Sci 2012; 1: 74-6. |
| 88 |
Wangchuk P, Giacomin PR, Pearson MS, et al. Identification of lead chemotherapeutic agents from medicinal plants against blood flukes and whipworms. Sci Rep 2016; 6: 32101.
DOI PMID |
| 89 |
Liu M, Panda SK, Luyten W. Plant-based natural products for the discovery and development of novel anthelmintics against nematodes. Biomolecules 2020; 10: 425-6.
DOI URL |
| 90 |
Soares AM, Lopes SG, et al. Anthelmintic activity of Leucaena leucocephala protein extracts on Haemonchus contortus. Rev Bras Parasitol Vet 2015; 24: 396-401.
DOI URL |
| 91 |
Kanojiya D, Shanker D, Sudan V, et al. Anthelmintic activity of Ocimum sanctum leaf extract against ovine gastrointestinal nematodes in India. Res Vet Sci 2015; 99: 165-70.
DOI URL |
| 92 |
Szewczuk VD, Mongelli ER, Pomilio AB. In vitro anthelmintic activity of Melia azedarach naturalized in Argentina. Phytother Res 2006; 20: 993-6.
PMID |
| 93 |
Ameen S, Azeez O, Baba Y, et al. Anthelmintic Potency of Carica papaya seeds against Gastro-intestinal Helminths in Red Sokoto goat. Ceylon J Sci 2018; 47: 137-41.
DOI URL |
| 94 | Al-Shaibani I, Phulan M, Arijo A, et al. Anthelmintic activity of Nigella sativa L, seeds on gastrointestinal nematodes of sheep. Pak J Nematol 2008; 26: 207-18. |
| 95 | Pal P, Tandon V. Anthelmintic efficacy of Flemingia vestita (Fabaceae) genistein-induced alterations in the ultrastructure of the tegument in the cestode, Raillietina echinobothrida. J Parasit Dis 2010; 22: 104-9. |
| 96 |
Hayes D, Angove MJ, Tucci J, et al. Walnuts (Juglans regia) chemical composition and research in human health. Crit Rev Food Sci Nutr 2016; 56: 1231-41.
DOI URL |
| 97 | Mali RG, Mahajan SG, Mehta AA. In vitro anthelmintic activity of stem bark of Mimusops elengi Linn. PhcogMag 2007; 3: 73-6. |
| 98 |
Aggarwal R, Kaur K, Suri M, et al. Anthelmintic potential of Calotropis procera, Azadirachta indica and Punica granatum against Gastrothylax indicus. J Parasit Dis 2016; 40: 1230-8.
DOI URL |
| 99 |
Jalalpure S, Alagawadi K, Mahajanashetti C, et al. In vitro anthelmintic property of various seed oils against Pheritima posthuma. Indian J Pharm Sci 2007; 69: 158.
DOI URL |
| 100 |
Jabbar A, Zaman MA, Iqbal Z, et al. Anthelmintic activity of Chenopodium album and Caesalpinia crista against trichostrongylid nematodes of sheep. J Ethnopharmacol. 2007; 114: 86-91.
PMID |
| 101 |
Afolayan M, Srivedavyasasri R, Asekun OT, et al. Phytochemical study of Piliostigma thonningii, a medicinal plant grown in Nigeria. Med Chem Res 2018; 27: 2325-30.
DOI PMID |
| 102 | Kiranmayi G, Ravishankar K, Priyabandhavi P. Phytochemical screening and in vitro comparative study of anthelmintic activity of Asparagus racemosus and Cucurbita maxima. J Pharm Res 2012; 5: 1545-7. |
| 103 |
Tangpu V, Temjenmongla K, Yadav AK. Anticestodal activity of Trifolium repens extract. Pharm Biol 2005; 42: 656-8.
DOI URL |
| 104 |
Hansson A, Zelada JC, Noriega HP. Reevaluation of risks with the use of Ficus insipida latex as a traditional anthelmintic remedy in the Amazon. J Ethnopharmacol 2005; 98: 251-7.
PMID |
| 105 |
Ayaz E, Gökbulut C, Coşkun H, et al. Evaluation of the anthelmintic activity of pumpkin seeds (Cucurbita maxima) in mice naturally infected with Aspiculuris tetraptera. J Pharmacognosy Phytother 2015; 7: 189-93.
DOI URL |
| 106 |
Bairwa R, Sodha R, Rajawat B. Trachyspermum ammi. Phcog Rev 2012; 6: 56.
DOI URL |
| 107 |
Williams AR, Ramsay A, Hansen TV, et al. Anthelmintic activity of trans-cinnamaldehyde and A-and B-type proanthocyanidins derived from cinnamon (Cinnamomum verum). Sci Rep 2015; 5: 14791.
DOI PMID |
| 108 | Dwivedi G, Bairagi M, Rawal D, et al. Anthelmintic activity of Myristica fragrans (Nutmeg) extract. Res J Pharm Bio Chem Sci 2011; 2: 315-8. |
| 109 |
Yan H, Haiming S, Cheng G, et al. Chemical constituents of the roots of Inula helenium. Chem Nat Compd 2012; 48: 522-4.
DOI URL |
| 110 |
Muthee J, Gakuya D, Mbaria J, et al. Ethnobotanical study of anthelmintic and other medicinal plants traditionally used in Loitoktok district of Kenya. J Ethnopharmacol 2011; 135: 15-21.
DOI PMID |
| 111 |
Olounladé P, Azando E, Hounzangbé-Adoté M, et al. In vitro anthelmintic activity of the essential oils of Zanthoxylum zanthoxyloides and Newbouldia laevis against Strongyloides ratti. Parasitol Res 2012; 110: 1427-33.
DOI PMID |
| 112 |
Souza M, Bevilaqua CM, Morais SM, et al. Anthelmintic acetogenin from Annona squamosa L. Seeds. An Acad Bras Ciênc 2008; 80: 271-7.
DOI URL |
| 113 | Bhinge SD, Hogade MG, Chavan C, et al. In vitro anthelmintic activity of herb extract of Eclipta prostrate L. against Pheretima posthuma. Asian J Pharm Clin Res 2010; 3: 229-30. |
| 114 | Karumari RJ, Sumathi S, Vijayalakshmi K, et al. Anthelmintic efficacy of Sesbania grandiflora leaves and Solanum torvum fruits against the nematode parasite Ascaridia galli. Am J Ethno Med 2014; 1: 326-33. |
| 115 | Nirmal S, Gagare P, Dighe S, et al. Anthelmintic activity of some existing polyherbal Ayurvedic formulations. Pharmacologyonline 2008; 3: 76-9. |
| 116 | Gajalakshmi S, Vijayalakshmi S, Devi RV. Pharmacological activities of Catharanthus roseus. Int J Pharm Bio Sci 2013; 4: 431-9. |
| 117 |
Maji AK, Banerji P. Chelidonium majus L.(Greater celandine)-A review on its phytochemical and therapeutic perspectives. Int J Herb Med 2015; 3: 10-27.
DOI URL |
| 118 |
Kozan E, Küpeli E, Yesilada E. Evaluation of some plants used in Turkish folk medicine against parasitic infections for their in vivo anthelmintic activity. J Ethnopharmacol 2006; 108: 211-6.
PMID |
| 119 |
Barrau E, Fabre N, Fouraste I, et al. Effect of bioactive compounds from Sainfoin (Onobrychis viciifolia Scop.) on the in vitro larval migration of Haemonchus contortus: role of tannins and flavonol glycosides. Parasitology 2005; 131: 531-8.
PMID |
| 120 | Saddiqe Z, Khalid S, Maimoona A. In vitro antelmintic activity of extracts of Withania Somnifera. J Nat Appl Sci Pakistan 2019; 1: 89-97. |
| 121 |
Eguale T, Tilahun G, Debella A, et al. In vitro and in vivo anth-elmintic activity of crude extracts of Coriandrum sativum against Haemonchus contortus. J Ethnopharmacol 2007; 110: 428-33.
PMID |
| [1] | LIU Xueling, MA Kun, TAO Wenhua, XU Zhongkun, LIU Gang, HU Chunyan, MAO Weiwei, GU Chang, GUO Qi. Natural products for treatment of premature ovarian failure: a narrative review [J]. Journal of Traditional Chinese Medicine, 2023, 43(3): 606-617. |
| [2] | PENG Wan, NI Hengfan, GUO Dale, DENG Yun, DAI Manyun. Farnesoid X receptor regulators from natural products and their biological function [J]. Journal of Traditional Chinese Medicine, 2023, 43(3): 618-626. |
| [3] | XUE Nannan, JIA Liyang, LI Qiyan. Potential of natural medicines for treatment of osteoporosis: a narrative review [J]. Journal of Traditional Chinese Medicine, 2023, 43(1): 198-204. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||